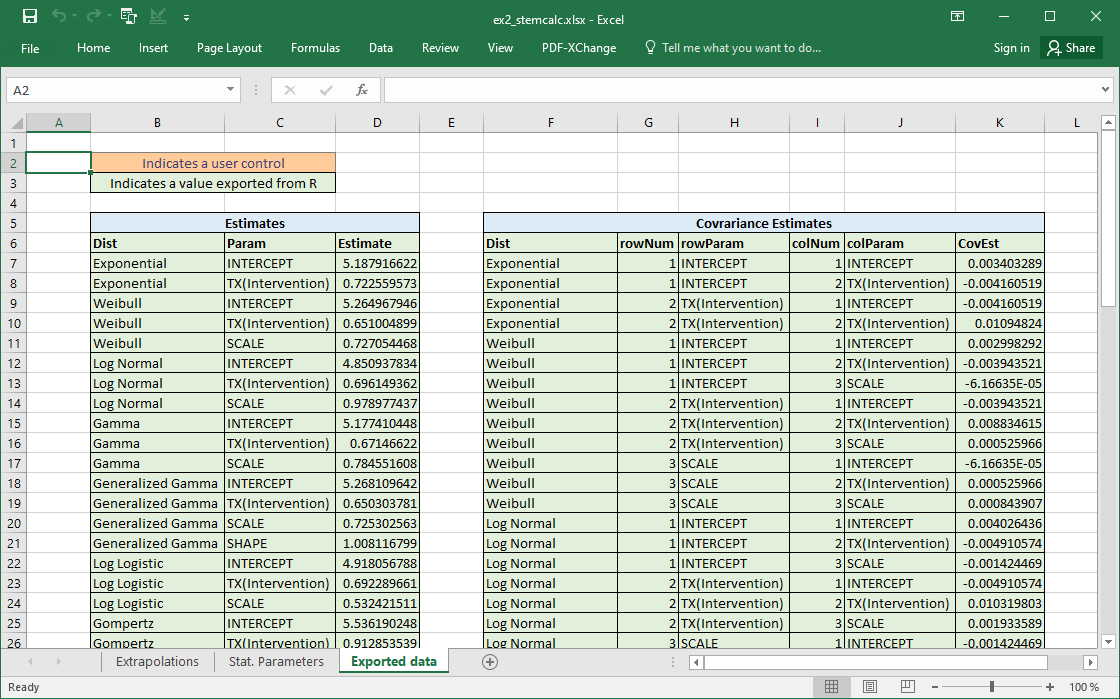
Excel example 2 - STEM compatibility
Roche
2023-05-02
Excel_example_ex2.RmdIntroduction
This vignette describes how to work with the included example excel templates that are compatible to the survival models estimated with flexsurvPlus. These examples are deliberately simple and are intended to illustrate calculations in excel rather than as a basis for a real economic model. In this example the basic calculations needed to extrapolate survival are illustrated. This example is using the STEM bacward compatibility formulas.
Set up packages and data
Generate the data
To perform survival analyses, patient level data is required for the survival endpoints. In this example, we analyze progression-free survival (PFS). For more details on these steps please refer to the other vignettes.
# make reproducible
set.seed(1234)
# used later
(simulation_seed <- floor(runif(1, min = 1, max = 10^8)))
#> [1] 11370342
(bootstrap_seed <- floor(runif(1, min = 1, max = 10^8)))
#> [1] 62229940
# low number for speed of execution given illustrating concept
n_bootstrap <- 10
adtte <- sim_adtte(seed = simulation_seed)
head(adtte)
#> USUBJID ARMCD ARM PARAMCD PARAM AVAL AVALU
#> 1 1 A Reference Arm A PFS Progression Free Survival 108 DAYS
#> 2 2 A Reference Arm A PFS Progression Free Survival 150 DAYS
#> 3 3 A Reference Arm A PFS Progression Free Survival 372 DAYS
#> 4 4 A Reference Arm A PFS Progression Free Survival 73 DAYS
#> 5 5 A Reference Arm A PFS Progression Free Survival 137 DAYS
#> 6 6 A Reference Arm A PFS Progression Free Survival 103 DAYS
#> CNSR
#> 1 0
#> 2 0
#> 3 0
#> 4 0
#> 5 0
#> 6 0
# subset PFS data and rename
PFS_data <- adtte %>%
filter(PARAMCD == "PFS") %>%
transmute(USUBJID,
ARMCD,
PFS_days = AVAL,
PFS_event = 1 - CNSR
)Fitting the models
More information about each function can be used by running the code ?runPSM or viewing the other vignettes.
Bootstrap the estimated parameters
As described in other vignettes we can use boot to
explore uncertainty.
Converting to STEM format
As described in other vignettes we can use convSTEM
function to transform the parameterisations for backwards compatibility
to the SAS macro model formulas.
stemdata <- convSTEM(x = psm_PFS_all, samples = boot_psm_PFS_all, use = "complete.obs")Exporting to Excel
Once the values are calculated we can export to Excel. The following code prepares two tibbles that can be exported. One containing the main estimates. A second containing the covariance matrices.
main_estimates <- stemdata$stem_param %>%
dplyr::transmute(Dist, Param, Estimate)
cov_estimates <- stemdata$stem_cov %>%
dplyr::transmute(Dist, rowNum, rowParam, colNum, colParam, CovEst)
# can preview these tables
main_estimates %>%
head() %>%
pander::pandoc.table()
#>
#> -------------------------------------------
#> Dist Param Estimate
#> ------------- ------------------ ----------
#> Exponential INTERCEPT 5.188
#>
#> Exponential TX(Intervention) 0.7226
#>
#> Weibull INTERCEPT 5.265
#>
#> Weibull TX(Intervention) 0.651
#>
#> Weibull SCALE 0.7271
#>
#> Log Normal INTERCEPT 4.851
#> -------------------------------------------
cov_estimates %>%
head() %>%
pander::pandoc.table()
#>
#> ---------------------------------------------------------------------------------
#> Dist rowNum rowParam colNum colParam CovEst
#> ------------- -------- ------------------ -------- ------------------ -----------
#> Exponential 1 INTERCEPT 1 INTERCEPT 0.003403
#>
#> Exponential 1 INTERCEPT 2 TX(Intervention) -0.004161
#>
#> Exponential 2 TX(Intervention) 1 INTERCEPT -0.004161
#>
#> Exponential 2 TX(Intervention) 2 TX(Intervention) 0.01095
#>
#> Weibull 1 INTERCEPT 1 INTERCEPT 0.002998
#>
#> Weibull 1 INTERCEPT 2 TX(Intervention) -0.003944
#> ---------------------------------------------------------------------------------
# the following code is not run in the vignette but will export this file
# require(openxlsx)
# wb <- openxlsx::createWorkbook()
# openxlsx::addWorksheet(wb, sheetName = "Exported data")
# openxlsx::writeDataTable(wb, sheet = "Exported data", main_estimates, startRow = 2, startCol = 2)
# openxlsx::writeDataTable(wb, sheet = "Exported data", cov_estimates, startRow = 2, startCol = 3+ncol(main_estimates))
# openxlsx::saveWorkbook(wb, file = "export_data_ex2.xlsx", overwrite = TRUE)The Excel model
Included with the package is an example Excel file called
ex2_stemcalc.xlsx. This can be extracted using the below
code (not run). It can also be found in the github repository at https://github.com/Roche/flexsurvPlus/tree/main/inst/extdata
installed_file <- system.file("extdata/ex2_stemcalc.xlsx", package = "flexsurvPlus")
installed_file
#> [1] "/usr/local/lib/R/site-library/flexsurvPlus/extdata/ex2_stemcalc.xlsx"
# not run but will give you a local copy of the file
# file.copy(from = installed_file, to ="copy_of_ex2_stemcalc.xlsx")This illustrates how all the included survival models can be extrapolated in Excel.
Stat. Parameters tab
This contains intermediary calculations needed when using the STEM parameterisation. This includes Cholesky decomposition of the calculated covariance matrix to implement PSA which are not needed when using the boot strap samples directly.
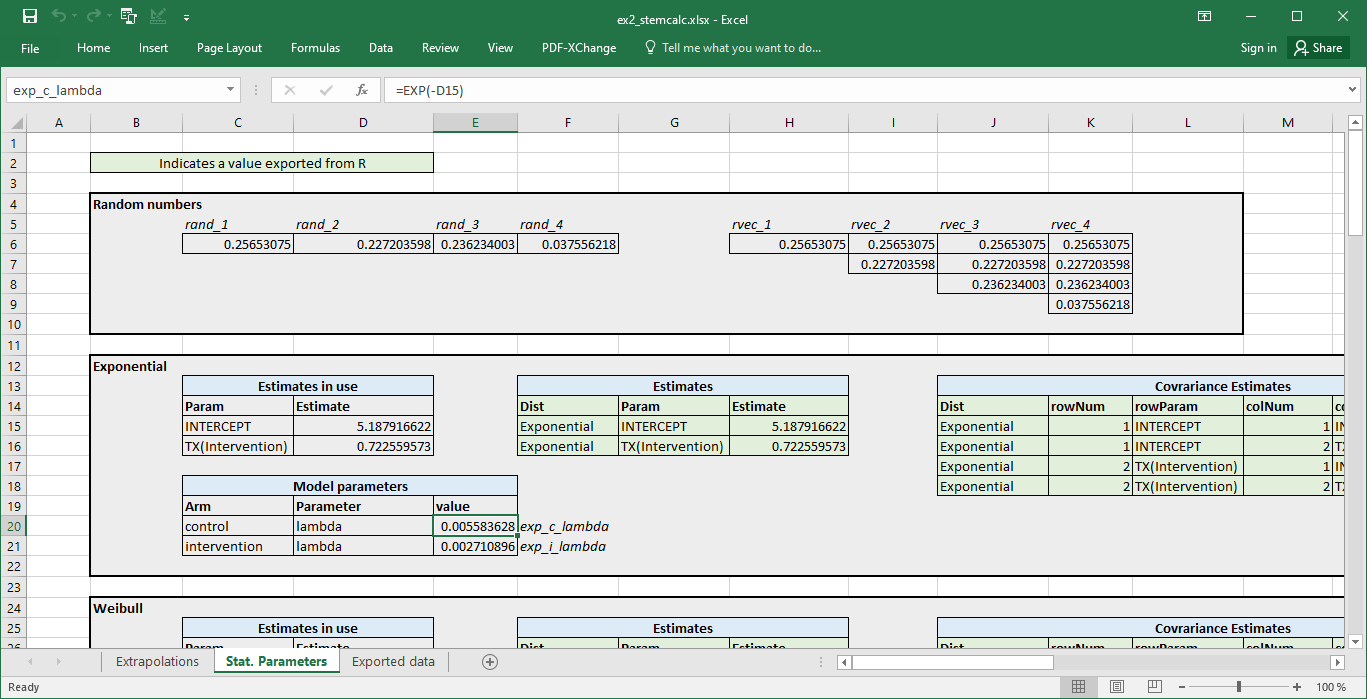
Stat. Parameters tab
Extrapolations tab
This contains example calculations to extrapolate survival.
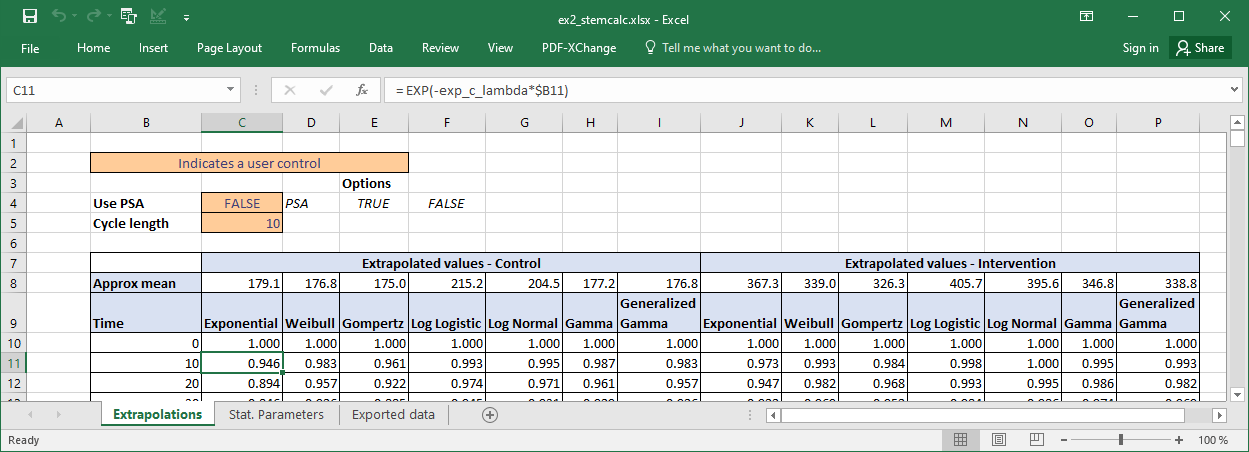
Extrapolations tab
We can compare the approximate estimates of mean survival with those calculated in R. As the excel model only goes until time t=2000 we can more directly compare to the estimates of restricted mean survival time (rmst) until this time.
means_est <- psm_PFS_all %>%
summaryPSM(type = c("mean", "rmst"), t = 2000)
# match to selected model in screenshot
means_est %>%
dplyr::filter(Model == "Common shape") %>%
tidyr::pivot_wider(
id_cols = c("Strata", "Dist"),
names_from = c("type"),
values_from = "value"
) %>%
dplyr::arrange(Strata, Dist) %>%
pander::pandoc.table()
#>
#> --------------------------------------------------
#> Strata Dist mean rmst
#> -------------- ------------------- ------- -------
#> Intervention Exponential 368.9 367.3
#>
#> Intervention Gamma 346.8 346.8
#>
#> Intervention Generalized Gamma 338.8 338.8
#>
#> Intervention Gompertz 326.3 326.3
#>
#> Intervention Log Logistic 459.4 405.7
#>
#> Intervention Log Normal 414.2 395.6
#>
#> Intervention Weibull 339 339
#>
#> Reference Exponential 179.1 179.1
#>
#> Reference Gamma 177.2 177.2
#>
#> Reference Generalized Gamma 176.8 176.8
#>
#> Reference Gompertz 175 175
#>
#> Reference Log Logistic 229.9 215.2
#>
#> Reference Log Normal 206.5 204.5
#>
#> Reference Weibull 176.8 176.8
#> --------------------------------------------------